Archived News
TB-MBLA Quarterly Newsletters are back again!
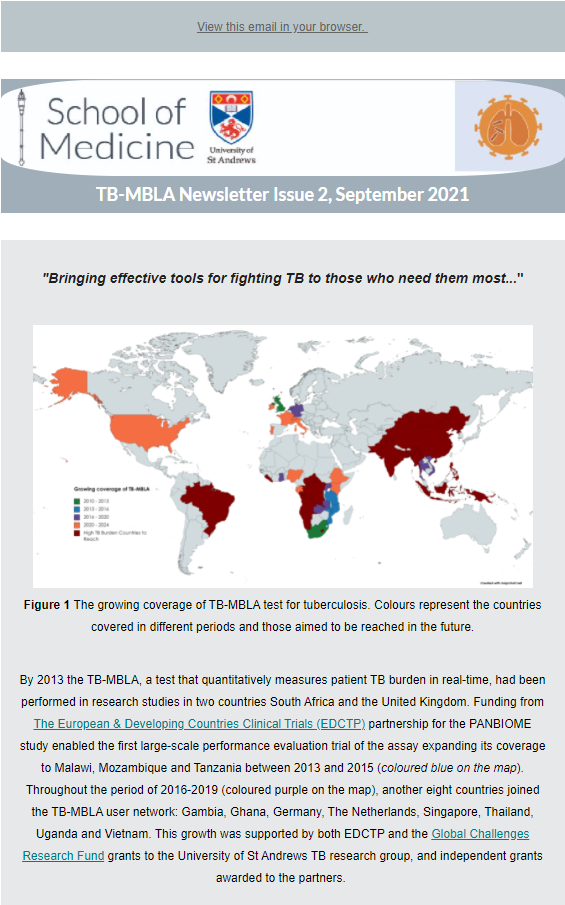
One of the focuses of the many clinical studies we conduct and participate in is the successful implementation of novel biomarkers in the clinical trial workflow to evaluate their performance and feasibility compared to standard microbiology practice. TB-MBLA has been a promising molecular tool that simplifies TB treatment monitoring by significantly reducing the turn- around time of quantitative bacterial load results. In doing so, patients’ responses to therapy can be followed real-time and with higher precision. The TB-MBLA research & implementation team led by Dr Wilber Sabiiti at the University of St Andrews is proud to have established a TB-MBLA User Group that consists of both research and industrial partners using and testing the performance of the assay across the world. With the purpose of maintaining the connection and communication with the members of the group, the idea of a TB-MBLA newsletter had arisen in previous years. The latest issue of the TB-MBLA Newsletter was published in June 2023.
SimpliciTB Strategic African network meeting – sustaining research capacity, Dar es Salaam, Tanzania 28th-30th September 2022
Our collaborators from the Global South assembled from NIMR-Mbeya, KCRI, TASK IHI, KIDH, KCRI, NIMR-Mbeya, NIMR-Mwanza, TASK, CERMEL, UCT, Wits Health, AURUM, and INSA gathered in Dar es Salaam on the 28th – 30th September.
Speaking via video link, Professor Stephen Gillespie opened the conference “Once upon a time in a country far far away: TB research in the 1990s”. This presented an overview of tuberculosis research in 1990 and how the collaboration had participated in a very significant change in research capacity through global collaboration in (PanACEA, EDCTP, TB Alliance link). Through a summary of our collaborative research: clinical phase 1-3 trials, and monitoring TB treatment response with molecular techniques TB-MBLA, he outlined how the research dynamic has changed together with some observations for the conference and the future.
One of the SimpliciTB Capacity Development aims is to strengthen research capacity to include the development of research leadership in sub-Saharan Africa. The SimpliciTB network has organized research capacity leadership into 4 workstreams addressing the training of various cadres involved in clinical trials, biomarkers, clinical trials, operational research, and community engagement measurement and evaluation. Implementation of workstreams activities prepares the Sub-Saharan Africa investigators to take leadership roles in the near future.
Dr Stella Mpagama was endorsed at the meeting as the African lead to coordinate colleagues to focus on simplifying TB treatment and apply regulatory phase III/IV trials to improve the capacity of sites. Validating TB diagnostics and drugs and future investment in infrastructure to support TB research work. A particular focus will be to invest in recruiting and developing young investigators and providing a supportive network for administrators, finance officers, and grant managers.
PanACEA & SimpliciTB Annual Meeting, Mangochi, Malawi
20-22 September 2022
Finally, after 2 years due to COVID-19, the PanACEA and SimpliciTB family were reunited in Malawi, hosted by Dr Marriott Nliwasa, Kamuzu University of Health Sciences at Makokola Retreat, Mangochi. The hospitality and organisation of all of the teams made it a very successful and enjoyable meeting.
PanACEA
The first day started with a variety of workshops being held on, Finance (Rutger Spoor), Statistics (Patrick Philips), Microbiology (Tim McHugh) and Pharmacology Adherence Technology (Marion Sumari-de Boer). The Tuesday afternoon, we unofficially started the first part of the General Assembly on the occasion of the 2022 Annual meeting. This part of the meeting was chaired by Marriott Nliwasa and the stage was set for the scientific presentations of all PhD and Master students who receive or have received grants from PanACEA. At the end of the afternoon, we were able to experience 2 presentations of the work packages with an overview of clinical trials HIGHRIF 1, BTZ043 and PHENORIF.
Wednesday 21st September Professor Martin Boeree (Radboudumc) and Dr Marriott Nliwasa (Kamuzu University Health Sciences, Malawi) officially opened the Annual General Meeting and welcomed all the delegates.
Dr Norbert Heinrich and Professor Martin Boeree provided an overview on the status and preparations of clinical trials SUDOCU and STEP2C. Next up was a full programme from the expertise cores – sponsor responsibility & trial management, Drug management, clinical pharmacology and trials design and statistics, microbiology, biomarkers, data management, and genetics all of whom gave a comprehensive update on their progress followed by engaging question and answer sessions.
Rutger Spoor and Iris Spelier provided an update on finance and communications, followed by a discussion on PANACEA 2 Past-Present-Future and Next steps!
The final session on Day 3 focussed on consortium governance and was co-chaired by Dr Nyanda Ntinginya (NIMR-Mbeya), Johann De Bruyn (TASK) and Stellah Mpagama (KIDH) discussing the future of PanACEA 3, in particular the North South and South-South roles, support within the consortium and consensus on collaborations for the future.
The final item on Thursday was a very constructive contribution by Norbert Heinrich on:
Kapiri
Malawi (one of poorest countries in the world) also faces some problems such as climate warming and lack of water. In cooperation with locals, trees are being planted to capture a huge amount of carbon dioxide. And since 2019, 4 boreholes have been realized. More information www.kapiri.de

SimpliciTB
Community Engagement (CE), Measurement & Evaluation Workshop –
20th September 2022
Here we built engagement capacity in clinical research, focussing on Good Participatory Practices (GPP) experiences and plans for a youth development programme. Importantly, we developed indicators for monitoring and evaluation impact of CE.
22nd September 2022
Dr Derek Sloan, Professor Stephen Gillespie and Julie Anderson (University of St Andrews) opened the meeting, welcoming international colleagues, reflecting on achievements and future goals. Rutger Spoor (Radboudumc) provided a finance update.
We welcomed Leandra Lombard (TB Alliance, South Africa) who provided a progress update on the SimpliciTB Trial and capacity development at sites.
Professor Timothy McHugh (University College London) provided a presentation on “Microbiology – beyond outcome data”.
The goal of Capacity Development is to support local sites to conduct clinical trials, upgrading existing infrastructure and promoting leadership.
- Dr Wilber Sabiiti and Dr Leticia Muraro Wilber (University College London) updated on Biomarker training and procurement of phase 1 and phase 2 equipment for the new trial sites.
- Drs Erica Sanga, Christine Laidson (NIMR-Mwanza, Tanzania) and Zani de Wit (UCT-LI, South Africa) summarised the Community Engagement activities, the Youth Health Ambassador Programme and updated delegates on the Monitoring & Evaluation programme.
- Dr Stella Mpagama and Dr Alphonce Liyoyo (Kibong’oto Infectious Diseases Hospital, Tanzania) introduced and updated on the progress of OptiRiMoxTB a new phase III multi-centre clinical trial, workshop sessions to discuss trial preparation were also held. Tanzania is leading the new trial, with mentorship from University of St Andrews and Radboudumc, strengthening research capacity in Gabon, Malawi & Mozambique.