Mackenzie Institute of Early Diagnosis > Research > Biophotonics
Light Sheet Microscopy
Led by Kishan Dholakia
In light sheet microscopy, we illuminate samples with a broad sheet of light rather than point by point scanning. This leads to faster image acquisition and lower sample exposure, thus leading to less “light damage”. Such imaging can lead to new insights for studies in neuroscience, diseases of the mind (dementia) and developmental biology. In turn this may shed light on numerous biological processes including the development of disease. In particular, we harness the non-diffracting properties of Airy beams in our light sheet microscopes to increase the field of view deeper into the sample. This has applications for histopathology and early diagnosis.
- Widefield light sheet microscopy using an airy beam combined with deep-learning super-resolution, S. Corsetti, P. Wijesinghe, P. B. Poulton, S. Sakata, K. Vyas, C. S. Herrington, J. Nylk, F. Gasparoli and K. Dholakia, OSA Continuum 3, 1068-1083 (2020)
- Light sheet fluorescence microscopy for Neuroscience, S. Corsetti, F. J. Gunn-Moore and K. Dholakia, Journal of Neuroscience Methods 319, 16 (2019)
- Light sheet Microscopy with acoustic sample confinement, Z. Yang, K. L. Cole, Y. Qiu, I. M. L. Somorjai, P. Wijesinghe, J. Nylk, S. Cochran, G. C. Spalding, D. A. Lyons, and K. Dholakia, Nat. Commun. 10, 669 (2019)
- Three photon light sheet fluorescence microscopy, A. Escobet-Montalbán, F. M. Gasparoli, J. Nylk, P. Liu, Z. Yang and K. Dholakia, Opt. Lett. 43, 5484 (2018)
- Light-sheet Microscopy with attenuation-compensated propagation-invariant beams, J. Nylk, K. McCluskey, M. A. Preciado, M. Mazilu, Z. Yang, F. J. Gunn-Moore, S. Aggarwal, J. A. Tello, D. E. K. Ferrier and K. Dholakia, Science Advances 4, eaar4817 (2018)
- Light-sheet microscopy using an airy beam, T. Vettenburg, H. I. C. Dalgarno, J. Nylk, C. Coll-Lladó, D. E. K. Ferrier, T. Čižmár, F. J. Gunn-Moore and K. Dholakia , Nature Methods 11, 541-544 (2014)
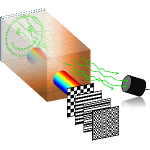
TRAFIX Microscopy
Led by Kishan Dholakia
The light-scattering properties of tissue make it incredibly hard to record images from deep within biological samples. We approach this challenge by implementing a scheme that requires no a priori information about the medium nor its properties. Exploiting temporal focusing, patterned illumination and single-pixel detection in our innovative scheme, we obtain wide-field two-photon images from deep within samples. The illumination intensity can be reduced further by optimising the basis in which the sequential illumination patterns are encoded.
- Optimal compressive multiphoton imaging at depth using sinlge-pixel detection, P. Wijesinghe, A. Escobet-Montalbán, M. Chen, P. R. T. Munro and K. Dholakia, Opt. Lett. 44, 4981 (2019)
- Wide-filed multiphoton imaging through scattering media without correction, A. Escobet-Montalbán, R. Spesyvtsev, M. Chen, W. Afshar Saber, M. Andrews, C. S. Herrington, M. Mazilu and K. Dholakia, Science Advances 4, eaau1338 (2018)
- Wide-field 3D optical imaging using temporal focusing for holographically trapped microparticles, R. Spesyvtsev, H. Rendall and K. Dholakia, Opt. Lett. 40, 4847 (2015)
Ultraviolet Light to Prevent Airborne Transmission of Viruses
Led by Kenny Wood
We have built a large interdisciplinary research team to study the safety and efficacy of ultraviolet light to prevent the spread of airborne bacteria and viruses. Our collaboration includes physicists, astronomers, virologists, biologists, radiation safety experts, and optics researchers from NHS Ninewells, the Universities of St Andrews, Dundee, Leeds, Heriot Watt University and Columbia University in New York, and local companies Fluid Gravity Engineering in St Andrews and Ten Bio in Dundee.
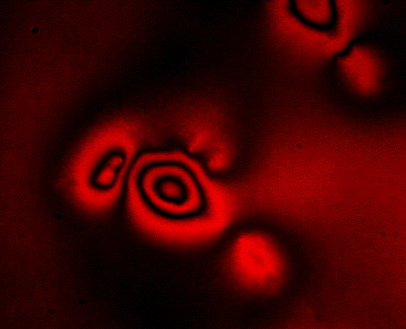
ERISM Cell Force Mapping
Led by Malte Gather
Biological cells continuously exert forces on their environment and these forces are key to many processes in health and disease, including cell growth, tissue formation, would healing and the invasion of cancer cells into health tissue. However, current methods to measure cellular force have a number of limitations which have so far prevented routine measurements of cellular force and use of force measurements in high throughput assays.With Elastic Resonator Interference Stress Microscopy (ERISM) we have developed a robust and non-destructive method to image the forces associated with various cell–substrate interactions. By exploiting optical interference, ERISM detects cell-induced deformations of a substrate with unprecedented sensitivity.
- KIAA0319 influences cilia length, cell migration and mechanical cell–substrate interaction, R. Diaz, N. M. Kronenberg, A. Martinelli, P. Liehm, A. C. Riches, M. C. Gather, and S. Paracchini, Scientific Reports 12, 722 (2022)
- Real-time imaging of cellular forces using optical interference, A. T. Meek, N. M. Kronenberg, A. Morton, P. Liehm, J. Murawski, S. J. Powis, and M. C. Gather, Nature Communications 12, 3552 (2021)
- Willin/FRMD6 affects influences the cell mechanical phenotype and neuronal differentiation in mammalian cells by regulating ERK1/2 activity, N. M. Kronenberg, A. Tilston-Lunel, F. Goff, K. Dholakia, M. C. Gather, and F. J. Gunn-Moore, Frontiers in Cellular Neuroscience 14, 552213 (2020)
- Cortical cell stiffness is largely independent of substrate mechanics, J. Rheinlaender, A. Dimitracopoulos, B. Wallmeyer, N. M. Kronenberg, K. J. Chalut, M. C. Gather, T. Betz, G. Charras, and K. Franze, Nature Materials 19, 1019-1025 (2020)
- Direct measurement of vertical forces shows correlation between mechanical activity and proteolytic ability of invadopodia, E. Dalaka, N. M. Kronenberg, P. Liehm, J. E. Segall, M. B. Prystowsky, and M. C. Gather, Science Advances 6, eaax6912 (2020)
- Long-Term Imaging of Cellular Forces with High Precision by Elastic Resonator Interference Stress Microscopy, N. M. Kronenberg, P. Liehm, A. Steude, J. A. Knipper, J. G. Borger, G. Scarcelli, K. Franze, S. J. Powis, M. C. Gather, Nature Cell Biology 19, 864–872 (2017)